Period 2 & Period 5 |
Chemistry in the Earth System is the chemistry/earth science course example for the 3-course model. See more in the Chemistry in the Earth System video
All of this can be an overwhelming transition without a little guidance. Below is:
1. A summary of the different units and their general subjects.
2. Each segment has a link to the unit's specific components.
You can also access the link of the same document on the NGSS site by using the link to the San Diego County Department of Education below.
NGSS Chemistry in the Earth System summary
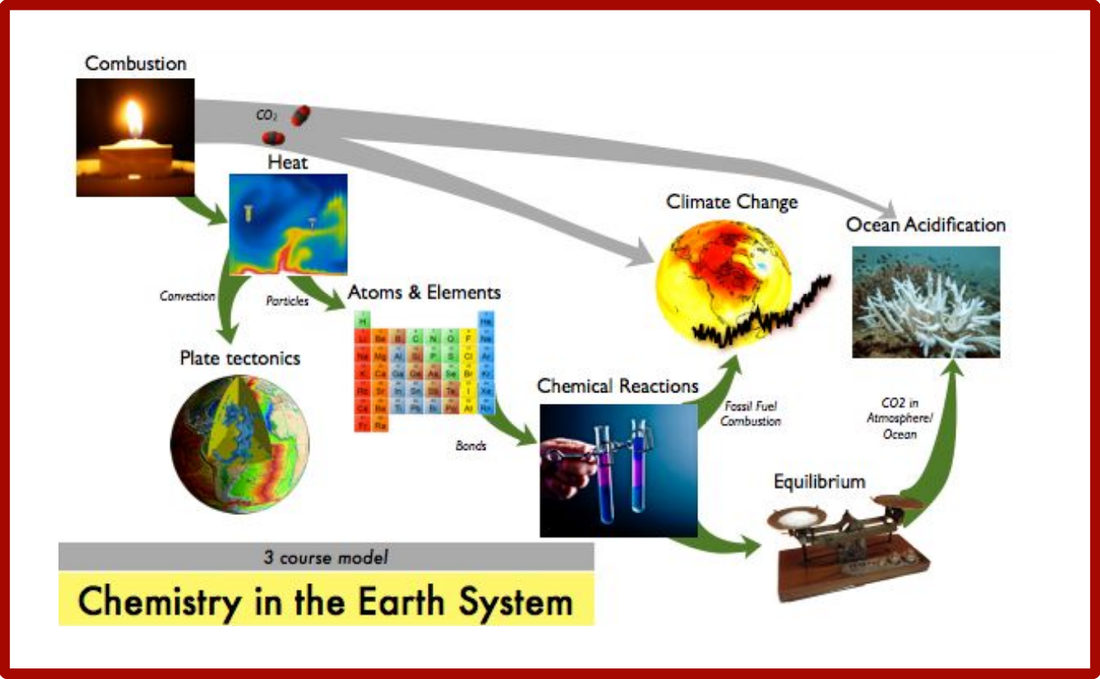
High School Chemistry in Earth Systems Course Storyline
Instructional Segment (Clickable Links Below) Description
SEMESTER 1
Unit 1
Combustion In this brief introductory unit, students investigate the amount of stored chemical potential energy in food. They make observations of material properties at the bulk scale that they will later explain in the atomic scale. The themes of combustion and CO2 tie together several of the Instructional Segments.
Unit 2
Heat and Energy in the Earth System Students develop models of energy conservation within systems and the mechanisms of heat flow. They relate macroscopic heat transport to atomic scale interactions of particles, which they will apply in later units to construct models of interactions between atoms. They use evidence from Earth’s surface to infer the heat transport processes at work in the planet’s interior.
Unit 3
Atoms, Elements, and Molecules Students recognize patterns in the properties and behavior of elements, as illustrated on the periodic table. They use these patterns to develop a model of the interior structure of atoms and to predict how different atoms will interact based on their electron configurations. They use chemical equations to represent these interactions and begin to make simple stoichiometric calculations.
SEMESTER 2
Unit 4
Chemical Reactions Students refine their models of chemical bonds and chemical reactions. They compare the strength of different types of bonds and attractions and develop models of how energy is stored and released in chemical reactions.
Unit 5
Chemistry of Climate Change Students develop models of energy flow in Earth’s climate. They revisit combustion reactions from IS1 to focus on emissions from fossil fuel energy sources. They apply models of the structures of molecules to explain how different molecules trap heat in the atmosphere. Students evaluate different chemical engineering solutions that can reduce the impacts of climate change.
Unit 6
Dynamics of Chemical Reactions and Ocean Acidification Students investigate the effects of fossil fuel combustion on ocean chemistry. They develop models of equilibrium in chemical reactions and design systems that can shift the equilibrium. Students conduct original research on the interaction between ocean water and shell-building organisms.
Subpages (6): Atoms, Elements, and Molecules Chemical Reactions Chemistry of Climate Change Combustion Heat and Energy in the Earth System The Dynamics of Chemical Reactions and Ocean Acidification
All of this can be an overwhelming transition without a little guidance. Below is:
1. A summary of the different units and their general subjects.
2. Each segment has a link to the unit's specific components.
You can also access the link of the same document on the NGSS site by using the link to the San Diego County Department of Education below.
NGSS Chemistry in the Earth System summary
High School Chemistry in Earth Systems Course Storyline
Instructional Segment (Clickable Links Below) Description
SEMESTER 1
Unit 1
Combustion In this brief introductory unit, students investigate the amount of stored chemical potential energy in food. They make observations of material properties at the bulk scale that they will later explain in the atomic scale. The themes of combustion and CO2 tie together several of the Instructional Segments.
Unit 2
Heat and Energy in the Earth System Students develop models of energy conservation within systems and the mechanisms of heat flow. They relate macroscopic heat transport to atomic scale interactions of particles, which they will apply in later units to construct models of interactions between atoms. They use evidence from Earth’s surface to infer the heat transport processes at work in the planet’s interior.
Unit 3
Atoms, Elements, and Molecules Students recognize patterns in the properties and behavior of elements, as illustrated on the periodic table. They use these patterns to develop a model of the interior structure of atoms and to predict how different atoms will interact based on their electron configurations. They use chemical equations to represent these interactions and begin to make simple stoichiometric calculations.
SEMESTER 2
Unit 4
Chemical Reactions Students refine their models of chemical bonds and chemical reactions. They compare the strength of different types of bonds and attractions and develop models of how energy is stored and released in chemical reactions.
Unit 5
Chemistry of Climate Change Students develop models of energy flow in Earth’s climate. They revisit combustion reactions from IS1 to focus on emissions from fossil fuel energy sources. They apply models of the structures of molecules to explain how different molecules trap heat in the atmosphere. Students evaluate different chemical engineering solutions that can reduce the impacts of climate change.
Unit 6
Dynamics of Chemical Reactions and Ocean Acidification Students investigate the effects of fossil fuel combustion on ocean chemistry. They develop models of equilibrium in chemical reactions and design systems that can shift the equilibrium. Students conduct original research on the interaction between ocean water and shell-building organisms.
Subpages (6): Atoms, Elements, and Molecules Chemical Reactions Chemistry of Climate Change Combustion Heat and Energy in the Earth System The Dynamics of Chemical Reactions and Ocean Acidification
https://sites.google.com/site/csunngssguide/high-school-model---3-course-model/three-course-model/chemistry-in-the-earth-system